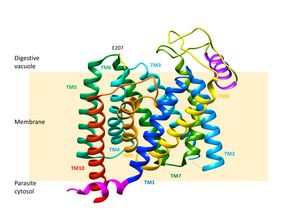
The emergence of drug-resistant strains of the malaria parasite Plasmodium falciparum has made the disease increasingly difficult to treat. One of the key proteins involved in drug resistance is the “Plasmodium falciparum chloroquine resistance transporter (PfCRT)”, a protein that is located in the membrane of the parasite's digestive vacuole. It expels anti-malarial drugs (like chloroquine) from the digestive vacuole so the drug loses its effectiveness. Like many transport proteins PfCRT is pH sensitive, but interestingly, it operates only at 60% of its maximal activity at pH 5.2, the pH of the digestive vacuole.
In a recent paper published in Nature Communications, a team of researchers, including UniSysCat PhD student Farzin Sohraby, Dr. Ariane Nunes Alves, Prof. Dr. Michael Lanzer and collaborators from Heidelberg University Hospital, investigated the pH dependence of PfCRT. Using a combination of different experimental and computational methods, the researchers were able to describe the structural and functional basis of the pH sensitivity displayed by PfCRT and link it to the transport cycle.
The authors performed a systematic mutational analysis of PfCRT with a focus on proton-accepting residues and found that a single residue, E207, serves as a critical pH sensor. But this feature is probably only a by-product: molecular dynamics (MD) simulations and kinetic studies suggest that the main function of E207 is in the transport cycle. It serves as a hydrogen acceptor for interactions with residue K80 via a salt bridge, thereby accelerating progression through the transport cycle. This would also explain why PfCRT hasn’t adapted to work at maximal activity at the pH of the digestive vacuole: the pH sensitivity is an intrinsic feature of how the transport process works in PfCRT.
This research provides the basis for designing new drugs targeting PfCRT that are more effective against drug-resistant strains of the malaria parasite.
Publication:
Berger, F., Gomez, G.M., Sanchez, C.P. et al. pH-dependence of the Plasmodium falciparum chloroquine resistance transporter is linked to the transport cycle. Nat Commun 14, 4234 (2023). https://doi.org/10.1038/s41467-023-39969-2