Organosilicon compounds, i.e., compounds that contain carbon-silicon bonds, are not found in nature but they can be artificially created. They are used in commercial products, for example, in antifoamers, caulks, or adhesives, and therefore play an important role in modern chemistry. However, some of those compounds, known as silanes, can be difficult to prepare and to further process in a selective manner.
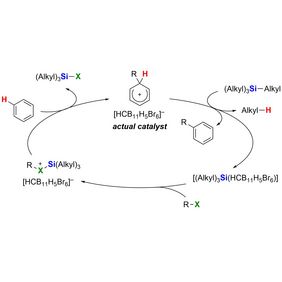
In their recent Nature publication “Arenium-ion-catalysed halodealkylation of fully alkylated silanes”, UniSysCat group leader Prof. Dr. Martin Oestreich describes with his coworkers, Dr. Hendrik Klare and Dr. Tao He, a novel top-down approach that effectively converts inert peralkylated silanes (silanes with four alkyl groups) into a diverse range of functionalized derivatives. This procedure has many advantages compared to previously reported bottom-up methods, as illustrated exemplarily within the synthesis of a silicon drug precursor.
While chlorosilanes such as Me4-nSiCln (n = 1 to 3) are a common starting point for the synthesis of silicon-containing molecules, fully alkylated silanes like Me4Si are usually considered synthetic dead ends. Despite that, by using the power of a catalytic reaction with an arenium-ion catalyst, the UniSysCat researchers were able to convert those inert silanes into more valuable forms. This happens in two steps in one pot: 1) protodealkylation and 2) halogenation.
This method could be used to improve industrial chemical production and serve as an entry into other reactions, such as the Tamao-Fleming oxidation, that is used, for example, in the synthesis of alcohol motifs.
Read the original publication at: https://www.nature.com/articles/s41586-023-06646-9