Catalytically Generated Meerwein’s Salt-Type Oxonium Ions for Friedel−Crafts C(sp2)−H Methylation with Methanol
by Tao He, Hendrik F. T. Klare, and Martin Oestreich
The group of UniSysCat group leader Martin Oestreich developed a new catalytic protocol which is important for the industrial methylation of aromatic compounds such as benzene and especially toluene, but also electron-deficient arenes under sustainable reaction condition via a Friedel−Crafts-type direct C(sp2)−H methylation.
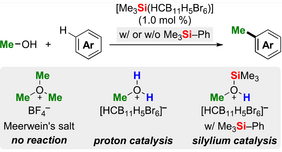
Counteranion-stabilized silylium or arenium ions, which form the so-called Meerwein’s salt-like oxonium ions initiate the underlying reaction utilizing methanol as the active methylating agents. Such silylated methyloxonium ions comprise stronger electrophiles than their protonated congeners, allowing the Friedel−Crafts alkylation to proceed more efficiently and at a lower reaction temperature, which is of high relevance for a sustainable industrial production. The regeneration of these superelectrophiles within the catalytic cycle is accomplished by the addition of a tetraorganosilane additive, i.e., trimethyl(phenyl)silane or tetraethylsilane. The latter release a silylium ion through protodesilylation by the Brønsted acidic "Wheland" intermediate, which acts as productive “proton-into-silylium ion” generator. This method allows even the C−H methylation of electronically deactivated aryl halides with methanol. In addition, this protocol can be also applied to nonactivated primary as well as π-activated benzylic alcohols and also dialkyl ethers could be used as alkylating agents.
Read more https://doi.org/10.1021/jacs.2c13341