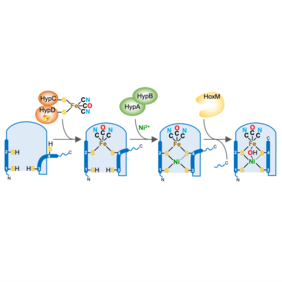
Stepwise assembly of the catalytic core of [NiFe]-hydrogenase
An international team involving the UniSysCat groups of Oliver Lenz, Christian Limberg, Peter Hildebrandt and Ingo Zebger, UniSysCat SAB member Serena DeBeer, and researchers from JASRI (Hyōgo, Japan) and DESY (Hamburg, Germany) deciphered the multistep incorporation process of the catalytic NiFe(CN)2(CO) cofactor into [NiFe]-hydrogenase by isolating previously uncharacterized protein intermediates trapped in different stages of the maturation process. These were analyzed using biochemical and a plethora of spectroscopic techniques.
Hydrogenases hold a promising future for hydrogen-based technologies and as a blueprint for chemically synthesized catalysts. The results of the study provide detailed insights into the assembly process of the intricate catalytic core of [NiFe]-hydrogenase. Moreover, the isolated intermediates serve as an ideal platform to study (semi-)artificial [NiFe]-hydrogenases equipped with synthetic iron complexes and Ni substitutes, potentially generating "chemzymes" with alternative catalytic functions.
This study has been published in Nature Chemical Biology: G. Caserta, S. Hartmann et al. Stepwise assembly of the active site of [NiFe]-hydrogenase. Nat. Chem. Biol. (2023). https://www.nature.com/articles/s41589-022-01226-w