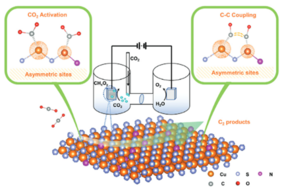
Strategies to achieve net zero carbon emissions must include ways to close the anthropogenic carbon cycle in the chemical sector through electricity-based, hence electrochemical valorization of CO2 into value-added compounds, such as ethylene (C2H4) and other multicarbon e-chemicals. In the direct electrocatalytic formation of such compounds from CO2, the elementary C–C coupling by CO dimerization is considered the rate-limiting step. In a new paper from UniSysCat researchers, a generalized surface structural design principle of asymmetric metal pair sites is proposed, explored, and experimentally tested in order to promote CO dimerization on surfaces. The electronic asymmetry of two copper metal centers result in distinct CO adsorption energies, which lowered C–C coupling energy barriers , yielding a significantly enhanced catalytic C2 faradaic efficiency.
Publication:
Asymmetric Copper-Sulphur Sites Promote C–C Coupling for Selective CO2 Electroreduction to C2 Products
L. Liang, L. Yang, T. Heine, A. Arinchtein, X. Wang, J. Hübner, J. Schmidt, A. Thomas, P. Strasser Adv. Energy Mater. 2024, 2304224
DOI: 10.1002/aenm.202304224