The UniSysCat groups of T. Braun, H. Dau, C. Limberg, M. Driess and K. Ray report the synthesis and characterization of an O2-derived four-coordinate [Fe2(μ-O)2]2+ complex and show that lower coordination number for the irons allows much more flexibility in the diamond core than proposed based on many other computational and experimental studies. The concept of building in flexibility by diminishing the metal coordination number has broader mechanistic significance because such changes within enzyme reaction cycles are used often by nature to drive respective catalysis. In this context, they provide spectroscopic insights into the structure of the so called " Q-intermediate" that is considered to be responsible for the conversion of methane to methanol in soluble methane monooxygenase enzyme.
Spectroscopic Properties of a Biologically Relevant [Fe (μ‐O) ] Diamond Core Motif with a Short Iron‐Iron Distance, D. Kass, S. Yao, K. B. Krause, T. Corona, L. Richter, T. Braun, S. Mebs, M. Haumann, H. Dau, T. Lohmiller, C. Limberg, M. Drieß, K. Ray, Angewandte Chemie International Edition 2023, 10.1002/anie.202209437
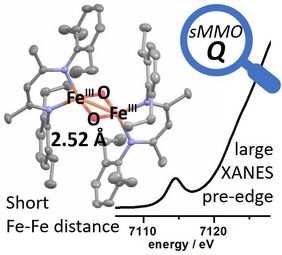
Diiron cofactors in enzymes perform diverse challenging transformations. The structures of high valent intermediates (Q in methane monooxygenase and X in ribonucleotide reductase) are debated since Fe−Fe distances of 2.5–3.4 Å were attributed to “open” or “closed” cores with bridging or terminal oxido groups. We report the crystallographic and spectroscopic characterization of a FeIII2(μ-O)2 complex (2) with tetrahedral (4C) centres and short Fe−Fe distance (2.52 Å), persisting in organic solutions. 2 shows a large Fe K-pre-edge intensity, which is caused by the pronounced asymmetry at the TD FeIII centres due to the short Fe−μ−O bonds. A ≈2.5 Å Fe−Fe distance is unlikely for six-coordinate sites in Q or X, but for a Fe2(μ-O)2 core containing four-coordinate (or by possible extension five-coordinate) iron centres there may be enough flexibility to accommodate a particularly short Fe−Fe separation with intense pre-edge transition. This finding may broaden the scope of models considered for the structure of high-valent diiron intermediates formed upon O2 activation in biology.