For a joint publication the five UniSysCat group of Shuang Li, Reinhard Schomäcker, Roel van de Krol, Peter Saalfrank and Arne Thomas teamed up to elucidate the impact of the orientation of a single functional group in a photocatalyst on its catalytic performance.
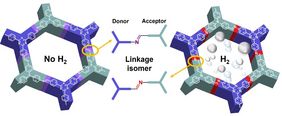
Constitutional (or structural) isomerism is a fundamental concept in chemistry. While it is well known that molecules, which are constitutional isomers possess different chemical and physical properties, isomers of extended organic structures like covalent organic frameworks (COFs) have not been investigated in the scientific literature so far.
Covalent organic frameworks (COFs) are porous crystalline materials composed of shape-persistent organic building units (linkers) connected through strong covalent linkages. While the nature of the linkers has been widely studied in the scientific literature, the orientation of the linkage between these linkers has not been considered as essential factor for the final properties of the material. The UniSysCat team however reasoned that the orientation of the linkage should be of high importance, especially for donor-acceptor (D-A) type COFs, currently vastly explored for different photocatalytic applications. Indeed, the prepared COF isomers showed substantial differences in their photophysical properties and consequently in their photocatalytic performance, exemplified in a ten times higher photocatalytic H2 evolution performance of one isomer over the other.
Constitutional isomerism of the linkages in donor–acceptor covalent organic frameworks and its impact on photocatalysis
J. Yang, S. Ghosh, J. Roeser, A. Acharjya, C. Penschke, Y. Tsutsui, J. Rabeah, T. Wang, S. Y. DjokoTameu, M.-Y. Ye, J. Grüneberg, S. Li, C. Li, R. Schomäcker, R. van de Krol, S. Seki, P. Saalfrank, A.Thomas Nat. Commun. 2022, 13, 6317 https://www.nature.com/articles/s41467-022-33875-9